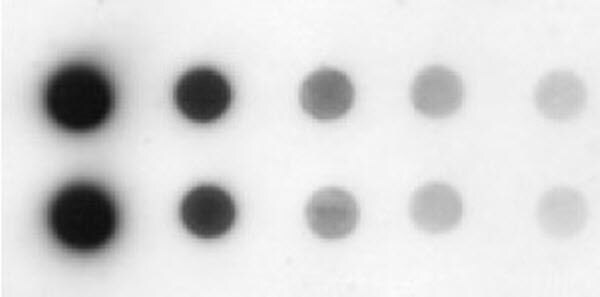
The FCIT, includes in addition to the latex reagent, a positive control, constituted by LPS O1 Ogawa, obtained, purified and drying under GMP conditions at the Finlay Vaccine Institute. To obtain the registration of this type of test is mandatory the qualitative and quantitative analysis of all its components to guarantee a good assay performance. Once the performance was demonstrated, the next step was to obtain the sanitary registration of the FCIT by the Cuban health regulatory authority, CECMED. The FCIT performance was evaluated in 210 samples of patients with a presumptive diagnosis of Cholera by the reference laboratory of the Institute of Tropical Medicine Pedro Kouri (IPK), showing sensitivity and specificity higher than 90%. cholera 01, which coupled to latex particles, allowed to obtain an Immunoagglutination test for the rapid diagnosis of the disease, so-called for us as Finlay Cholera Immunoagglutination Test (FCIT). At that time, the Finlay Vaccine Institute had already obtained a monoclonal antibody against LPS of V. In 2010, with the re-emergence of Cholera in Haiti, Cuba revitalized epidemiological surveillance for the timely diagnosis of this infection. Both systems have been used either individually or simultaneously and have revolutionized the diagnosis of the disease. The most recent methods are based on monoclonal antibodies (mAbs), coupled to: membranes (immunochromatography or test strips) or to latex particles (immunoagglutination). Some methods are based on polyclonal antibodies that allow identifying the bacteria directly from the primary isolation in culture medium. Several methods have been developed for the rapid diagnosis of V. So a rapid diagnostic test is a promising tool to detect cholera outbreaks in areas without laboratory infrastructure. Contradictorily, stool culture is the gold standard for confirmation and characterization of the outbreak strain, but it is time-consuming, requires trained technicians and a functional laboratory. The torpid evolution of the disease, together with its similarity with other ADDs and the high cost involved to implement epidemiological control measures, makes the rapid diagnosis of the disease, a fundamental pillar in the control of the illness. From the clinical point of view Cholera is characterized by secretory diarrhea, from moderate to severe, leading to rapid dehydration, that cause a considerable number of deaths. Although, more than 200 serogroups have been identified to date, only serogroups O1 and O139 are responsible for the epidemics that have occurred. Of the five FCIT positive control lots evaluated, four complied with 30% of the expected concentration, for an 80% effectiveness of the technique.Ĭonclusions: These results suggest that the quantitative Dot Blot using the mAbC anti Vibrio cholerae LPS O1, can be employed for the quantification of LPS in batches of the FCIT positive control, from the purification and production stages, as well as for the stability evaluation.Ĭholera is an acute diarrheal disease (ADD) caused by ingestion of food or water contaminated with bacillus Vibrio cholerae. A four-parameter fit model curve was obtained with R2 of 0.99. Results: The mAb was conjugated to the HRP efficiently with working dilutions range from 1:2500 to 1:10,000. The LPS concentration in positive control lots was calculated employed Ascent Software. The images were captured using the GS-800 densitometer and the spots density (Int/mm2) were calculated using the ImageJ software. The development of the reaction was carried out using SIGMAFAST™ DAB Tablet. For detection conjugated mAb-HRP was employed at dilution 1:5000. For capture Vibrio cholerae 569B Lipopolysaccharides from Sigma, was used as standard of the curve (40 μg/mL to 0.6 μg/mL) and five lots of FCIT positive control were applied as sample. Quantitation of LPS O1 was accomplish by quantity Dot Blot. Materials and Methods: Conjugation of mAb anti LPS O1 to peroxidase enzyme was carry out by periodate method. The objective of the work was to develop a quantitative Dot Blot to determine the LPS O1 concentration in FCIT Positive control, using a peroxidase-conjugated mAb. FCIT, includes a positive control (LPS O1 Ogawa), which quantification is mandatory to obtain the registration of the test. Finlay Vaccine Institute obtained an Immunoagglutination test for rapid diagnosis of Cholera (FCIT), based on a monoclonal antibody anti-LPS O1 coupled to latex particles. Contradictorily the gold standard test (stool culture) takes several days. Introduction: Rapid diagnosis is fundamental for epidemiological control of Cholera disease.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
